Electrophoretic painting, or e-coating as it may be referred to, is an advanced and sophisticated process that is used for the application of a semi-transparent and anti-corrosive layer as a coating on specific types of metals. But the difference between e-coating and other coating methods is that e-coating does not provide the material with an enamel-looking finish. This technique is applicable to several different types of metallic objects like vehicles and furniture. However, it is prevalent amongst jewelers. The reason is that it opens the possibility for unconventional colors to be incorporated in their designs while retaining the metallic shine of the pieces.
What Is E-Coating?
A combination of plating and painting is what e-coating can be defined as in simple terms. The metal piece is soaked in a paint emulsion surrounding water. Next, an electric current is passed through the solution containing resins and pigments for color. The resulting chemical reaction from the electric current leads to the adhesion of the paint onto the metal’s surface.
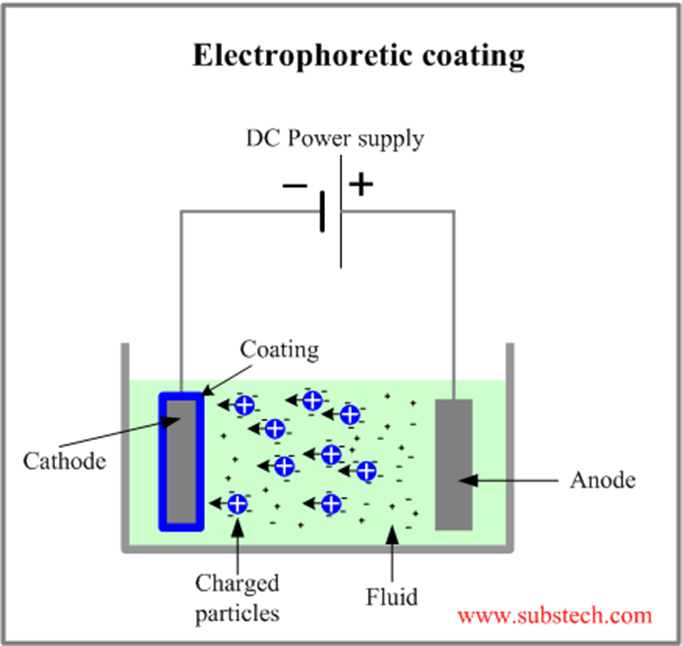
Afterward, the metal gets removed from the solution mix and allowed to rest to harden the coating while exposing it to high temperatures for a particular amount of time. Later, the metal goes through polishing to create a smooth finish on the surface.
Benefits of Using E-Coating
The pointers below explain the specific advantages for the jewelry industry amongst the countless that e-coating has to offer. Let’s have a look at them.

Types of E-Coating
Essentially, there are two types of e-coating, including anodic e-coating and cathodic e-coating.
Anodic E-Coating
In an anodic e-coating system, the part remains the anode, meaning that it is positively charged and draws in the negatively charged paint particles in the bath. Nevertheless, small traces of metal ions relocate from the part into the paint film while restricting the performance properties of these systems. So, in this system, the metal ions pollute the coating; therefore, this process isn’t broadly utilized because of the decreased corrosion resistance and durability.
Cathodic E-Coating
In cathodic e-coating systems, the part stays negatively charged, drawing in the positively charged paint particles. The polarization of the negative charge of the piece significantly reduces the chances of iron entering the cured paint film. Apart from that, it also upgrades the corrosion resistance and longevity and diminishes contamination in the baths. In other words, cathodic e-coat layers deliver enhanced performance with excellent resistance to corrosion. Along the same lines, suppose you are going to e-coat. In that case, this appears to be the best technique.
What to Consider Before Starting E-Coating?
The following are a few things to consider prior to moving towards the e-coating technique:
Type of Metal
E-coating is advantageous but not suitable for costume jewelry. The reason is that the process can cause the materials to burn. Also, uncharacteristic metals such as titanium and niobium are more complicated to e-coat because it remains considerably more challenging to make the color stick to the metal. It makes e-coating best to use on standard metals like gold, silver, and platinum.
Design Components
The e-coating process may not be suitable for pieces containing design components such as glass stones or enamel, as they might get abandoned during the e-coating process.
Color
Any color can be implemented through the e-coating process. However, the color will be tough to remove. Therefore, please select your color carefully so that you avoid regretting it later.
After these factors are considered, you can now prepare your item for e-coating.
The 6-Step Process
Polishing the Surface
The first step is to polish the metal surface to remove any irregularities present on it. It is regardless of the type of metal the item is being composed of. Impurities such as dirt, grease, and grime will also be removed using this process. Remember that rough textures produce uneven coatings.
Cleaning the Surface
Polishing may clean most impurities, but it may also result in metal filings and other residues. These may hinder the adhesion of the e-coat. In order to proceed with the e-coating, the surface should be cleaned properly. The surface cleaning solution to be used will be dependent on the type of metal the pieces are made of. An inorganic phosphate solution is typically the perfect choice for iron and steel. Conversely, alkaline cleaners are ideal for metals such as gold and silver.
The best tooling option is an ultrasonic cleaner. Mechanical vibrations by the tank create sound waves in the water or solution. Metal objects placed in the solution will be cleaned by bubbles created from the sound waves. These bubbles will efficiently clean even hard-to-reach points.
Rinsing
After being void of all dirt, debris, and scratches, the item is rinsed in distilled water and a neutralizer. It allows to clear off any remaining chemical residue from cleaning procedures. The process is repeated to ensure that no trace of chemicals or impurities remains on the metal. The more accurate this step is, the better the chances of adequate adhesion in the e-coating process.
Dipping in a Wetting Agent
For preventing bubbles from adhering to the item prior to submerging in the e-coating solution, the metal is dipped in a wetting agent, which is also known as a leveling agent, by some manufacturers. This agent diminishes the surface tension of the e-coating solution, which implies that its adhesion to the metal surface will be in a more cohesive manner. At the same time, this step also lowers the likelihood of any paint shortcomings on the final finished item.
Submerging In E-Coating Solution
After proper cleaning and application of the wetting agent, it is time to immerse the item in the e-coating solution. Again, the chemicals utilized in this solution may vary based on specific parameters, including the type of metal we are working with. It is crucial to note that the entire item should be submerged. Ultimately, it will bring about that the coating is even over the whole object, even on the spots that are hard to reach. A chemical reaction will result from the electrical currents passing through the e-coating solution, fusing the coating to the surface.
Cure the Coating
After the item is removed from the solution, it is taken to be baked in an oven or kiln. It is done to harden the coating of the metal and ensure durability. The process also adds a glossy finish. The temperature for this process varies based on the materials used in the e-coating solution. This step should not be rushed as it may adversely affect the quality of the final product.
Final Words
The benefit of e-coating is that it has revolutionized the fashion industry significantly by enabling artisans to make reliable and long-lasting jewelry in a broad scope of colors. The semi-transparent e-coat solutions allow the metal underneath to be still showcased. Eventually, it makes e-coating a favorite choice amongst jewelry makers who do not want their jewelry pieces to have a miserable and matte finish. The process is also eco-friendly compared to other coating techniques. The process, however, is highly technical and is best left to the experts only.

