Often, there are some questions frequently asked by machine shops and manufacturers of various part materials, including stainless steel, titanium, and tantalum. These incorporate questions such as “what is passivation,” “how does passivation work,” and “how to passivate stainless steel parts or components after machining operations?” Today in this article, we will investigate these questions, answering each to help you better understand the process. So, let’s get started.
Passivation of Stainless Steel
For preventing corrosion and rust, we use a typical finishing process known as passivation. In the case of stainless steel, we use nitric acid or citric acid in the passivation process to eliminate the free iron present on the surface. A defensive layer of oxide that is less chemically reactive with the air around it to prevent corrosion is formed by this chemical treatment. In turn, when stainless steel gets passivated, it will resist rusting and corrosion.
Meaning of Passivated Stainless Steel
For manufacturers, the industry standards such as ASTM A967 and AMS 2700 demonstrate the most commonly used standards to passivate stainless steel. As stated in ASTM A967, passivation is defined as the chemical treatment of the metal with a trivial oxidant, such as stainless steel with a nitric acid solution, while removing the free iron or other external matter.
Furthermore, ASTM A380 defines that passivation refers to removing exogenous iron or iron compounds from the stainless steel surface with the help of chemical dissolution – most generally through treatment with an acidic solution, which will eliminate the surface contamination but will not considerably influence the stainless steel itself. It further states that this process aims at enhancing the extemporaneous formation of the external protective film.
Benefits of Passivating Stainless Steel

After manufacturing and fabricating newly-machined stainless steel parts or components, it is often best to perform passivation. Some of the benefits it provides are:
- It protects the pieces from rust by creating a chemical layer over them.
- It increases the product’s lifespan and durability.
- The contamination present on the product’s surface is removed.
- The requirement of maintenance is reduced.
How Does Passivation Process Work?
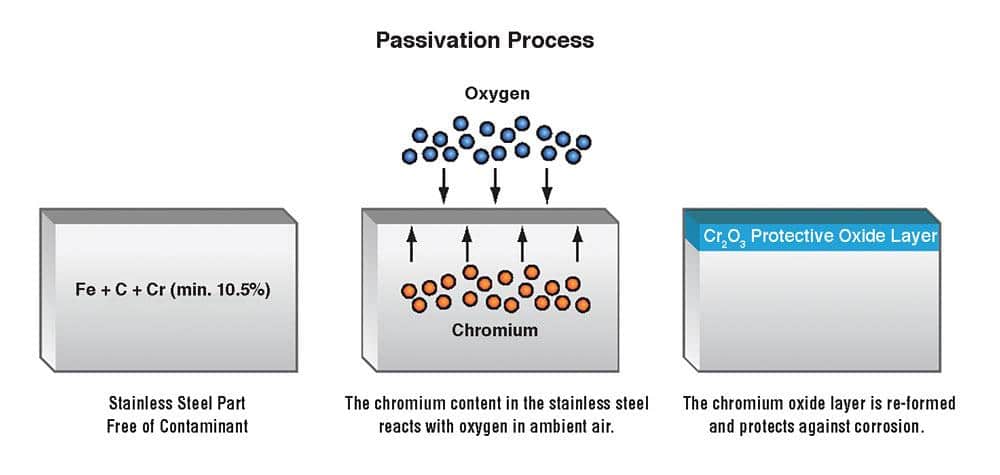
Typically, iron, nickel, and chromium are what stainless steel is composed of as it refers to an iron-based alloy. Moreover, the chromium present in the stainless steel is what gives it resistance against corrosion. Basically, chromium protects the iron from getting rusted by forming a layer of chromium oxide when exposed to oxygen present in the air. Meanwhile, the formation of the chromium oxide layer is enhanced and amplified by the passivation process.
The chromium stays intact while the free iron present on the surface gets dissolved when we immerse the stainless steel into an acid bath. The chromium forms a uniform layer on the surface with higher proportions than the underlying iron due to the acid dissolving all the free iron.
The chromic oxide layer forms over 24 to 48 hours when the stainless steel is exposed to oxygen present in the air after the acidic bath. The chromium oxide layer formed on the surface is thicker and provides more corrosion protection. No doubt, it is due to the high amount of chromium present at the surface in comparison to other metals. Also, opportunities for corrosion to take place are stopped as we remove the free iron present on the surface. Remember, it results in a protection against rust by the chemically non-reactive surface provided by the passive layer.
When Is Passivation Required?
After the processes such as grinding, welding, cutting, and other machining operations that influence stainless steel are performed, we then carry out the passivation process. However, passivation may seem unnecessary as stainless steel already resists corrosion in ideal conditions.
But then again, in practical situations, there might be some difficulties that can cause the oxide layer not to form correctly and not be able to protect against corrosion. These include:
- Unwanted material being present during manufacturing, such as grinding swarf, dirt, etc.
- The sulfides added to increase the machinability of stainless steel.
- Iron particles from cutting tools get inserted into the surface of stainless steel parts.
For acquiring a uniform corrosion-resistant surface, these impurities must be eliminated entirely. Undoubtedly, these problems can be corrected by performing the passivation process.
What Passivation Is NOT?
Not Electrolytic. Passivation is not an electrolytic process and does not rely on electrochemical reactions. Instead, passivation is a chemical treatment.
Not For Scale Removal. Passivation does not refer to the removal of oxide scale from machined parts after welding or heat treating.
Not a Coat of Paint. The surface color or appearance of stainless steel does not change when passivated. For items to be painted or powder-coated, passivation is not required.
How to Passivate Stainless Steel?
For passivating stainless steel, titanium, and different materials, many passivation specifications (AMS 2700, ASTM A967) are present for instructing on how to passivate properly. The chemical cleaning and passivation process phases, which are standard to almost all the specifications, are:
- Clean – Impurities such as oil and grease present on the surface should be removed.
- Passivate – Submerging the material into an acid bath containing nitric or citric acid.
- Test – To ensure the previous steps’ effectiveness, test the passivated stainless steel surface.
According to some specifications, sodium dichromate can also be added to the nitric acid bath to increase the speed of the formation of the oxide layer (passivation film). But sodium dichromate is a hexavalent chromium compound, which is highly toxic. Therefore, alternative processes are used to encourage oxygen formation at the metallic surface while the material is still submerged into the acidic bath. Examples of these alternatives include ultrasonic machines and citric acid.
The metal is immersed in the acid tank for typically 20 to 30 minutes. The temperature, however, can vary in a more considerable range, generally between 120 to 150 °F. It is because the temperature is mainly dependent on the grade of the stainless steel and the acid chemistry.
Steps for Passivating Stainless Steel Parts
For a passivation line to be put together, a process is required that will allow for the stainless steel to be both cleaned and passivated. Simultaneously, the most common and necessary steps involved in the process of the passivation of stainless steel are as follow:
- All contaminants such as oils and foreign materials are removed with the use of alkaline cleaning. Sodium hydroxide, Micro-90, and Simple Green are a few examples of the commonly used detergent cleaners in this process.
- DI (Deionized) or RO (Reverse Osmosis) water is used to perform a water rinse in high-precision industries.
- An immersion bath of nitric or citric acid is prepared to dissolve any free ions and sulfides on the surface completely. It also accelerates the formation of the passive oxide film.
- Water rinse again to clear off any remaining acid residue, usually with Deionized water in high-precision industries.
- Another water rinse, again preferably with Deionized water in high-precision industries.
- Now, the parts need to be dried out.
- While using a salt spray, chamber exposure, high humidity, or copper sulfate testing, the sample parts are tested for several specification standards and criteria.
What to Keep In Mind When Passivating Metals?
Passivation of metals can be termed as a method of controlled corrosion. As acid would typically work on metal, in passivation, the acid bath dissolves/corrodes the free iron present on the surface level in a uniform and well-ordered way. Nonetheless, if the process is not controlled correctly, a phenomenon called a “flash attack” could occur, leading to runaway corrosion. This flash attack results in the metal developing a dark and significantly etched surface, which is the type of corrosion that is meant to be prevented through the use of a passive layer.
But then again, a few steps can be taken to prevent this from happening.
Ensuring No Contaminants in Acid Solution
It is critical for preventing flash attacks. This remedy often involves refilling the acidic tank with a fresh solution regularly while avoiding contaminants in the acid bath solution. Another recommendation is the use of higher grades of water, such as RO or DI water, with relatively few chlorides compared to tap water. So, it may also prevent such problems as flash attacks.
Thorough Cleaning Of Metal Parts
The cleaning of the metal parts before the acid bath refers to yet another crucial process to act as a countermeasure for severe issues. Any impurity such as grease or cutting oil on the parts will form bubbles that may interfere with the whole process. Consider the use of a degreaser for such scenarios.
Multiple detergents separately or changing the current detergent could also work to ensure the part is free of all sorts of contaminants. Occasionally, thermal oxides from welding or heat treating might call for grinding or pickling to be removed before the process of passivation.
Being Careful About Mixing Grades of Stainless Steel
Stainless steel grades such as 300 series and 400 series should not be mixed in the acid bath simultaneously. The reason is that it might increase the probability of galvanic corrosion.


